Xenical or Orlistat. Some of you that have been around for a while have likely heard of Xenical or Alli as another low-dose version that was available over the counter. Many of you that have been struggling with your weight for some time have maybe even been on it. Xenical next to phentermine is one of the oldest weight-loss medications currently on the market, gaining FDA approval in 1999.
So why haven’t you talked about it until now?
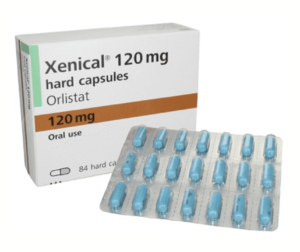 Well before we dive into that backdoor issue let’s review what Xenical or orlistat, the generic name, is. Xenical is what we call a lipase inhibitor. When it comes to our digestion system, in order for our body to absorb nutrients from the food we eat, it needs to break food down into its smallest components.
Well before we dive into that backdoor issue let’s review what Xenical or orlistat, the generic name, is. Xenical is what we call a lipase inhibitor. When it comes to our digestion system, in order for our body to absorb nutrients from the food we eat, it needs to break food down into its smallest components.
This process starts in our mouth where our teeth pulverize the food and add a few enzymes to start breaking things down. It then goes to our stomach where the food basically enters a washing machine full of acid; things get broken down and dissolved even further. From there, it enters our intestines where the food or what I believe is called chyme, if I remember my high school biology correctly, then passes to the small intestine.
In the small intestine, the chyme meets more enzymes and chemicals that break it down even further from large complex molecules as seen below to small individual molecules in order for our body to then absorb them and utilize them for bodily functions. One of the enzymes that help to break down fats is called lipase!
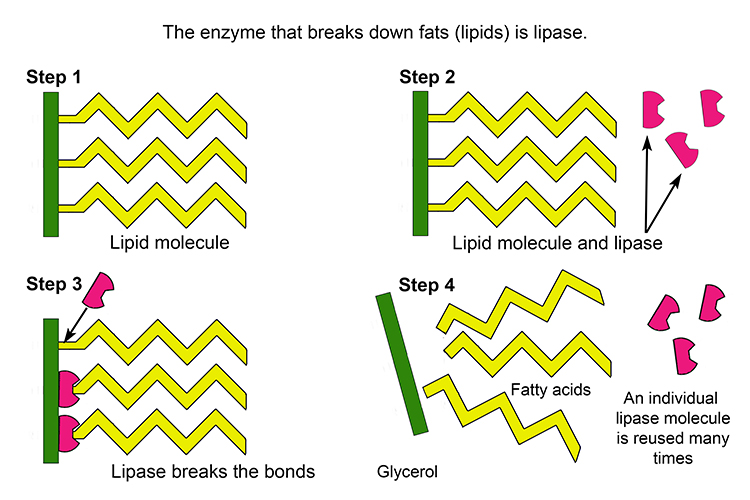
A lipase inhibitor like Xenical does just as the name implies – it inhibits lipase. If lipase is inhibited it is then unable to break down fats. If fats are not broken down they are not absorbed, therefore reducing the number of calories our body takes up! Of course, this leads to a reduced enough calorie intake that an individual creates a calorie deficit (calories in < calories out) and weight loss will occur.
In the case of Xenical, this was found to be true. An interesting 2-year study by Davidson and friends looked at using Xenical first with a focus on weight loss during year 1 then in year 2 the study transitioned to preventing weight regain in patients with Obesity.
In the first 52 weeks, there were two groups and both were on a calorie-restricted diet:
- Placebo – placebo capsule 3x/day with meals
- Xenical – Xenical 120mg 3x/day with meals
In year 2, we see some changes in terms of the groups and all participants were then put on a weight maintenance diet:
- Placebo – This group continued as is.
- Xenical – this group was re-randomized into 3 other groups:
- Placebo – switched from Xenical to placebo capsule 3x/day with meals.
- Xenical 60mg – dose decreased but still took medication 3x/day with meals
- Xenical 120mg – continued on current Xenical dosage 3x/day with meals.
And what kind of results did Davidson and friends find?!
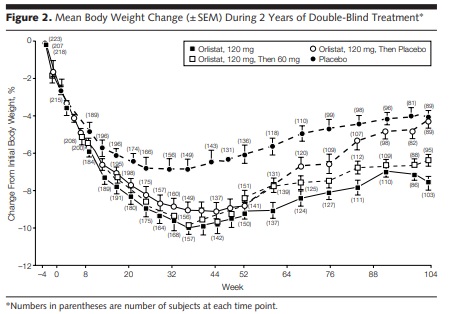
In the first 52 weeks, the Xenical group lost more weight on average compared to placebo 8.8% vs. 5.8% from baseline, respectively. As expected, when we look at year 2, the individuals that stayed on Xenical 120mg regained less weight on average compared to the Xenical 60mg and placebo groups, 3.2kg vs. 4.26kg vs. 5.63kg, respectively. The graph below gives you a much better visual representation of what actually happened! Further, the high-dose Xenical group saw improvements in LDL or bad cholesterol and insulin levels.
An additional study called the XENDOS trial looked at individuals with diabetes and obesity. It was found that Xenical may help to reduce the incidence of developing diabetes or at least delay the time to development which can certainly be considered a win.
So the results seem pretty solid; am I finally going to mention why I haven’t talked about Xenical thus far?!
Yes, please tell me about Xencal!
 Absolutely, let’s get into the ‘bulk’ of the matter!
Absolutely, let’s get into the ‘bulk’ of the matter!
I was aiming for a poop joke there.
I don’t think it quite landed.
Anyways, what do you think happens when fat is ultimately not broken down or absorbed when you take Xenical? That’s right, it ends up in your bowel movements. Needless to say, for a large number of people this is not a fun time.
In fact, Davidson and friend’s trial had a dismal completion rate, with less than 50% of the participants completing the entirety of the study. The main side effects reported were flatus with discharge, oily spotting, fecal urgency, fatty/oily stool, oily evacuation, and increased defecation. Basically, these individuals could not trust a fart, had to buy new underwear and couldn’t leave the house due to oily diarrhea.
Unfortunately, these side effects are quite common, occurring in at least 25% of individuals. So hopefully that explains why I am apprehensive about using this medication unless of course you perhaps have an issue with constipation or if one of my enemies comes to me for weight management support. Thus far in my 5+ years of practice, I have only prescribed Xenical once.
Final thoughts from Dr. Dan
So that is Xenical. It is still an agent that is available and could be effective for supporting weight management. However, it quite literally is a sh*tty alternative to the other agents that are out there.
As a final note, besides the side effects, you need to be mindful of any fat-soluble vitamins D, E, A, and K as their concentrations can be reduced while taking this medication. Xenical may also interact with certain medications, and the fact you need to take it 3x/day with meals is kind of an additional pain in the ass.
Anywho, hopefully, that answers your questions about Xenical and provides a healthy dose of poop-related humour!
Until next my friends, always remember that small tweaks lead to massive peaks.
– Dr. Dan
References
Torgerson, J. S., Hauptman, J., Boldrin, M. N. & Sjöström, L. XENical in the prevention of diabetes in obese subjects (XENDOS) study: a randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care 27, 155–161 (2004).
Davidson, M. H. et al. Weight Control and Risk Factor Reduction in Obese Subjects Treated for 2 Years With Orlistat: A Randomized Controlled Trial. JAMA 281, 235–242 (1999).